Pharnext Announces Financing of €11 Million Through a Capital Raise Subscribed by Existing Shareholders and a Convertible Bonds Issued to European Investors; European Investors Also Provide Indication of Interest for an Additional Amount of up to €25 Million in Convertible Bonds
- €6 million capital raise priced at market with warrants subscribed by two existing shareholders,
- €5 million convertible bonds issued to European investors,
- European investors have also provided written indication of strong interest for the potential of additional €25 million in similar convertible bonds, depending on future market conditions
PARIS, France, 10:00 p.m. CET, February 4, 2021 – Pharnext SA (FR0011191287 - ALPHA) (the “Company”), an advanced late-stage clinical biopharmaceutical company pioneering new approaches to developing innovative drug combinations based on big genomics data and artificial intelligence using its PLEOTHERAPY™ platform, today announced a financing of €11 million through the combination of a €6 million capital raise subscribed by two existing shareholders by way of issuance of new ordinary shares (the “New Shares”) with one warrant attached (together with the New Shares, the “ABSA”) and €5.0 million of convertible bonds (the “ConvertibleBonds”) subscribed by European investors, who have expressed strong written interest for a potential additional €25 million in similar convertible bonds over the next 30 months, depending on future market conditions.
“We are pleased to announce this capital raise which demonstrates the strong continued support of our existing shareholders”, said David Horn Solomon, Chief Executive Officer. “This funding will extend our cash runway to advance PXT3003 into the pivotal phase III trial in Charcot-Marie-Tooth disease Type 1A”.
“We have structured the financing to include a convertible bond in order to manage dilution for existing shareholders and still provide funding for Pharnext to get through important near-term milestones, while preserving the flexibility to increase the size of the convertible bonds facility later if needed, given the strong interest received from European investors”, said Peter Collum, Chief Financial Officer and Chief Business Officer of Pharnext.
This capital raise did not and will not require the publication of a prospectus subject to the approval of the Autorité des marchés financiers (“AMF”).
Use of Proceeds
The proceeds from the financing will fund operations through several key milestones expected within the first half of 2021, including the initiation of the pivotal phase III clinical trial for PXT3003 in Charcot-Marie-Tooth disease Type 1A in Q1 2021 as well as top-line data from the interim analysis of our ongoing long-term extension study from the first phase III clinical trial for PXT3003 in Charcot-Marie-Tooth disease Type 1A.
Terms of the ABSA
The ABSA have been issued by the Company’s board of directors on February 3, 2021, in accordance with the 19th resolution of the General Shareholders’ Meeting as of July 17th, 2020 (the “General Meeting”), without shareholders’ preferential subscription rights, to CB Lux and Lohas, two of the main shareholders of the Company and whose representative or person controlling are represented on the Board of Directors.
The issue price of one ABSA is € 3.42, issue premium included (the “Issue Price”), with New Share priced at no discount and representing the volume weighted-average price of the Company’s shares over the last three trading days (the “3-day VWAP”). The Issue Price of an ABSA, including the theoretical value of the warrant attached thereto (“BSA”), represents a total 14.74% discount to the 3-day VWAP, consistent with the 19th resolution of the General Meeting.
Terms of the BSA
One BSA is attached to each New Share.
Two BSAs entitle their holder to subscribe to one new ordinary share of the Company, at a price of € 4.45 representing a 30% premium to the Issue Price.
The BSAs may be exercised at any time within 60 months of their issuance. In the event all BSAs are exercised a total number of 877,193 additional ordinary shares of the Company will be issued, generating total proceeds of approximately € 3.9 million.
The theoretical value of each BSA, assuming a volatility of 45% and based on Issue Price, is equal to € 0.50 under Black & Scholes model, representing a 14.74% discount to the 3-day VWAP.
The BSAs will be immediately detached from the New Shares upon issuance and will not be listed.
Terms of the Convertible Bonds
The issuance of the Convertible Bonds has been approved by the Company’s board of directors on February 3, 2021, in accordance with the 19th resolution of the General Meeting, without shareholders’ preferential subscription rights, to Vester Finance and two other European investors.
The Convertible Bonds were issued at a price equal to 95% of their nominal value. The total nominal amount of the Convertible Bonds is € 5,473,685 and the net proceeds for the Company € 5,00,000.75 after deduction of the 5% discount on the nominal value and the commission fees. The Convertible Bonds will bear no interest and will mature on February 4th, 2024. The Convertible Bonds will be converted into ordinary shares at any time at a subscription price per share (the “Conversion Price”) equal to the lowest of (i) €4.25 and (ii) 93% of a lowest daily VWAP over the 15 trading-day period preceding such conversion, providing that the Conversion Price may in no event be lower than the 3-day VWAP prior to the conversion date discounted by 30%.
New shares resulting from the conversion of Convertible Bonds will be fully fungible with existing ordinary shares and will enjoy the same rights.
In connection with the issuance of the Convertible Bonds, the Company has entered into a standstill agreement, which restricts the issuance of additional instruments giving access to new shares with a variable subscription price as long as any Convertible Bonds are still outstanding and unless the subscribers of such instruments accept to enter into a lock up agreement for the duration of the Convertible Bonds.
The Convertible Bonds will not be the subject of an application for admission to trading on Euronext Growth. The issue will not give rise to a prospectus submitted for approval to the AMF.
The transaction was advised and structured by Vester Finance, which also subscribed for Convertible Bonds.
Potential additional €25 million in similar Convertible Bonds
The Convertible Bonds subscribers have provided written indication of strong interest for a potential additional €25 million in similar convertible bonds over the next 30 months, upon terms to be mutually agreed, depending on future market conditions, including liquidity factors, but not-milestones based.
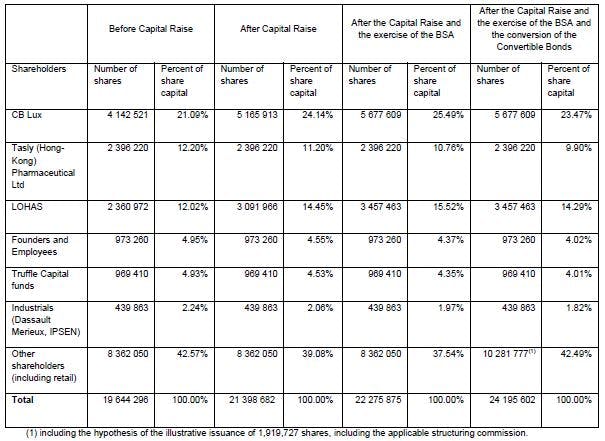
The Company’s shareholding structure after the Capital Raise and the Convertible Bonds
Following the issuance of the ABSA, the Company’s total share capital will be € 213,986.82 composed of 21,398,682 ordinary shares with a par value of €0.01, representing 108.93% of the total current share capital of the Company.
Lohas S.à.r.l, a company controlled by Mr. Pierre Bastid, himself a member of the Board of Directors of the Company, and CB LUX S.à.r.l, a company represented by Mr. Alexandre Berda, himself a member of the Board of Directors of the Company, holding respectively 12.02% and 21.09% of Pharnext’s share capital prior to the Capital Raise, have subscribed the Capital Raise for an aggregate amount of € 6,000,000. Following completion of the Capital Raise, they will own 14.45% and 24.14% respectively of the share capital of the Company.
On an illustrative basis, a shareholder holding 1% of the Company’s share capital (on a non-diluted basis) before the Capital Raise will hold:
- 0.92% of the Company’s shares after the Capital Raise; and
- 0.88% of the Company’s shares after the Capital Raise and the exercise of the BSA; and
- 0.81% of the Company’s shares after the Capital Raise, the exercise of the BSA and the conversion of all of the € 5,473,685 Convertible Bonds on the Conversion Price by using the lowest of the last 15 daily VWAP as of today of €3.06, resulting in the illustrative issuance of 1,919,727 shares.
Risk Factors
The Company draws the public’s attention to the risk factors related to the Company and its activities presented in section 3 of the universal registration document approved with the AMF under number R. 20. 029 on November 9, 2020 as well as in its annual periodic management reports and press releases, copies of which are available free of charge on the website of the Company (www.pharnext.com).
In addition, investors are invited to consider the following risks: (i) the market price for the Company's shares may fluctuate and fall below the subscription price of the shares issued pursuant to the Capital Raise, (ii) the volatility and liquidity of the Company's shares may fluctuate significantly, (iii) sales of Company’s shares may occur on the market and have a negative impact on the market price of the shares, and (iv) the Company’s shareholders could undergo a potentially material dilution resulting from any future capital increases that are needed to finance the Company.
About Pharnext
Pharnext is an advanced clinical-stage biopharmaceutical company developing novel therapeutics for orphan and common neurodegenerative diseases that currently lack curative and/or disease-modifying treatments. Pharnext has two lead products in clinical development. PXT3003 completed an international Phase III trial with positive topline results for the treatment of Charcot-Marie-Tooth disease type 1A and benefits from orphan drug status in Europe and the United States. PXT864 has generated encouraging Phase II results in Alzheimer’s disease and will be advanced through partnerships. Pharnext has developed a new drug discovery paradigm based on big genomics data and artificial intelligence: PLEOTHERAPY™. Pharnext identifies and develops synergic combinations of drugs called PLEODRUG™. More information can be found at www.pharnext.com.
Pharnext is listed on the Euronext Growth Stock Exchange in Paris (ISIN code: FR0011191287).
Disclaimer
This press release contains certain forward-looking statements concerning Pharnext and its business, including in respect of timing of and prospects for clinical trials and regulatory submissions of the Company’s product candidates as well as a potential financing transaction, the use of proceeds therefrom and cash runway. Such forward-looking statements are based on assumptions that Pharnext considers to be reasonable. However, there can be no assurance that the estimates contained in such forward-looking statements will be verified, which estimates are subject to numerous risks including the risks set forth in Pharnext’s URD approved by the AMF on November 9, 2020 under number N° R. 20-029 as well as in its annual periodic management reports and press releases (copies of which are available on www.pharnext.com) and to the development of economic conditions, financial markets and the markets in which Pharnext operates. The forward-looking statements contained in this press release are also subject to risks not yet known to Pharnext or not currently considered material by Pharnext. The occurrence of all or part of such risks could cause actual results, financial conditions, performance or achievements of Pharnext to be materially different from such forward-looking statements. Pharnext disclaims any intention or obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise.]
This press release and the information that it contains do not constitute an offer to sell or subscribe for, or a solicitation of an offer to purchase or subscribe for, Pharnext shares in any country, including the United States. The Company’s securities may not be offered or sold in the United States absent registration or an exemption from registration; any public offering of securities to be made in the United States will be made by means of a prospectus that may be obtained from the issuer that will contain detailed information about the Company and management, as well as financial statements.