PXT3003
CMT1A is a demyelinating inherited peripheral neuropathy characterized by muscle weakness and atrophy in arms and legs, leading to progressive motor disability and sensory loss. CMT1A is caused by a duplication of the PMP22 gene, resulting in over-expression of the PMP22 protein and failure of the Schwann cells to produce normal myelin (sheath of neurons). The lack of normal myelin structure and function leads to abnormal peripheral nerve conduction, axonal loss and muscle atrophy.
PXT3003 is a novel fixed-dose synergistic combination of baclofen, naltrexone and sorbitol formulated as an oral solution given twice a day. The three individual components of PXT3003 were selected to downregulate the overexpression of PMP22 protein, leading to improvement of neuronal signaling in dysfunctional peripheral nerves that are an essential part of the pathophysiology of this disease. PXT3003 could also have a positive effect on other cellular types of the motor unit such as the axon (direct protection), neuromuscular junctions or muscle cells.
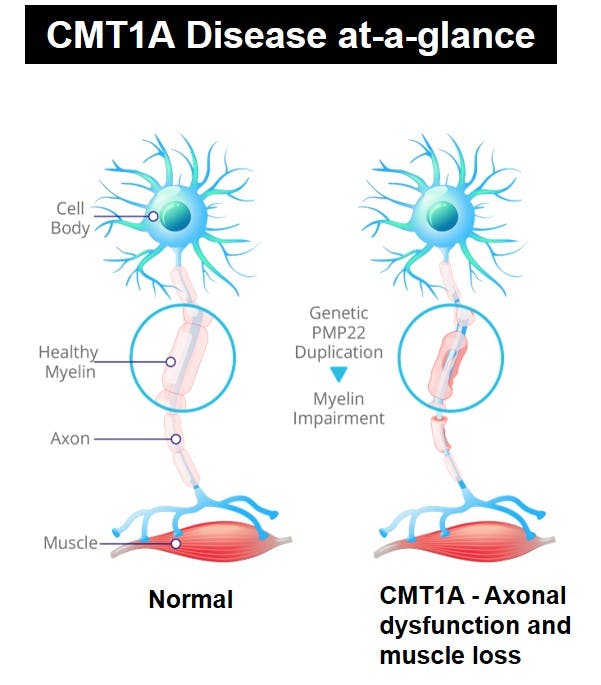
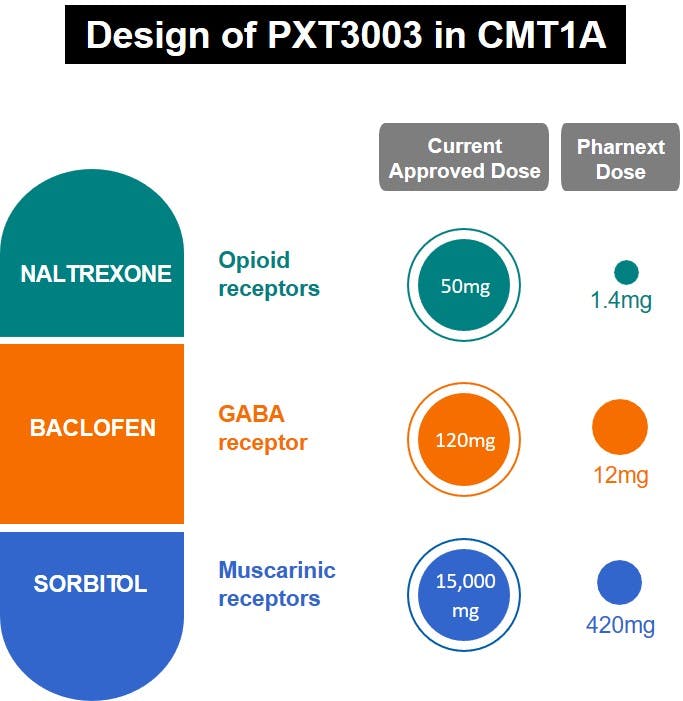
PXT3003 has shown promising and consistent results across preclinical and clinical studies in Phase 2 and Phase 3.
Over the preclinical phase, PXT3003 was evaluated in in vitro and in vivo rodent models of CMT1A. In two different studies, PXT3003 was able to inhibit the over-expression of the PMP22 gene and improve peripheral nerve myelination and motor/sensory impairments in transgenic CMT1A rats. Single components or dual combinations of PXT3003 active ingredients were not able to show such positive results both in vitro and in vivo models (Chumakov et al. OJRD 2014; Prukop et al. J Neurosci Res. 2020). These promising preclinical studies led to the launch of a clinical development program, that evaluated the potential clinical benefit and safety of PXT3003 in CMT1A patients.
The safety, tolerability and preliminary efficacy profile of PXT3003 given over 12 months was first characterized in a multicenter, randomized, double-blind, placebo-controlled Phase 2 study performed in France in 80 patients suffering from mild-to-moderate CMT1A. PXT3003 was found to be safe and well tolerated in all three doses tested. Encouraging efficacy data and dose effect were demonstrated by using the Overall Neuropathy Limitation Scale (ONLS), a validated scale that measures functional disability. Results from this study supported the design and initiation of the first Phase 3 trial.
The first Phase 3 study which further evaluated the efficacy and safety of PXT3003 was an international multicenter, randomized, double-blind, placebo-controlled, 15-month clinical trial. Two dose levels (highest dose used in the Phase 2 and twice that dose) of PXT3003 in comparison to placebo were tested in 323 mild-to-moderate CMT1A patients, enrolled at 29 centers across Europe, the United States and Canada from December 2015. Due to an unexpected formulation issue occurring in the high dose, this treatment arm was prematurely stopped in September 2017. A revised statistical analysis plan was developed to take into account the high number of missing data due to the discontinuation of patients in this treatment arm. In October 2018, Pharnext announced that analysis of the primary endpoint, ONLS (as advised by the FDA and EMA), from all investigated populations in the high-dose arm, suggested preliminary efficacy in humans. The study also further demonstrated the safety and tolerability of PXT3003.
Pursuant to this Phase 3 clinical trial, patients were eligible to continue treatment with PXT3003 in an open-label extension study. Results suggested an adequate safety profile over long-term use and sustained efficacy of PXT3003 in CMT1A patients after 6 years of total trial time. In December 2023, 120 patients enrolled in the first Phase 3 were still on treatment with PXT3003.
A second Phase 3 trial, requested by he FDA and EMEA, was launched to confirm the efficacy and safety of PXT3003. This trial, called PREMIER, was an international, multicenter, randomized, double-blind, placebo controlled, 15-month pivotal Phase 3 trial. One dose level of PXT3003 (high dose of the first Phase 3) was tested versus placebo in 387 mild-to-moderate CMT1A patients, enrolled in 52 centers worldwide from March 2021. In December 2023, Pharnext announced that analysis of the primary endpoint, ONLS scale which measures functional disability, did not confirm findings from previous clinical trials. Patients with mild-to-moderate CMT1A experienced improvement on both treatment and placebo, rather than the slow deterioration typical of CMT1A’s natural progression. Other data suggest no deterioration in the condition of patients under treatment, requiring further analysis. The trial reaffirmed the high safety profile of the treatment, already established in previous studies.
Patients completing the PREMIER trial had the opportunity to continue treatment with PXT3003 in an open-label extension study (OLE-PREMIER trial) to generate additional long-term data. In December 2023, 315 patients enrolled in PREMIER trial were still on treatment with PXT3003..
