PXT864
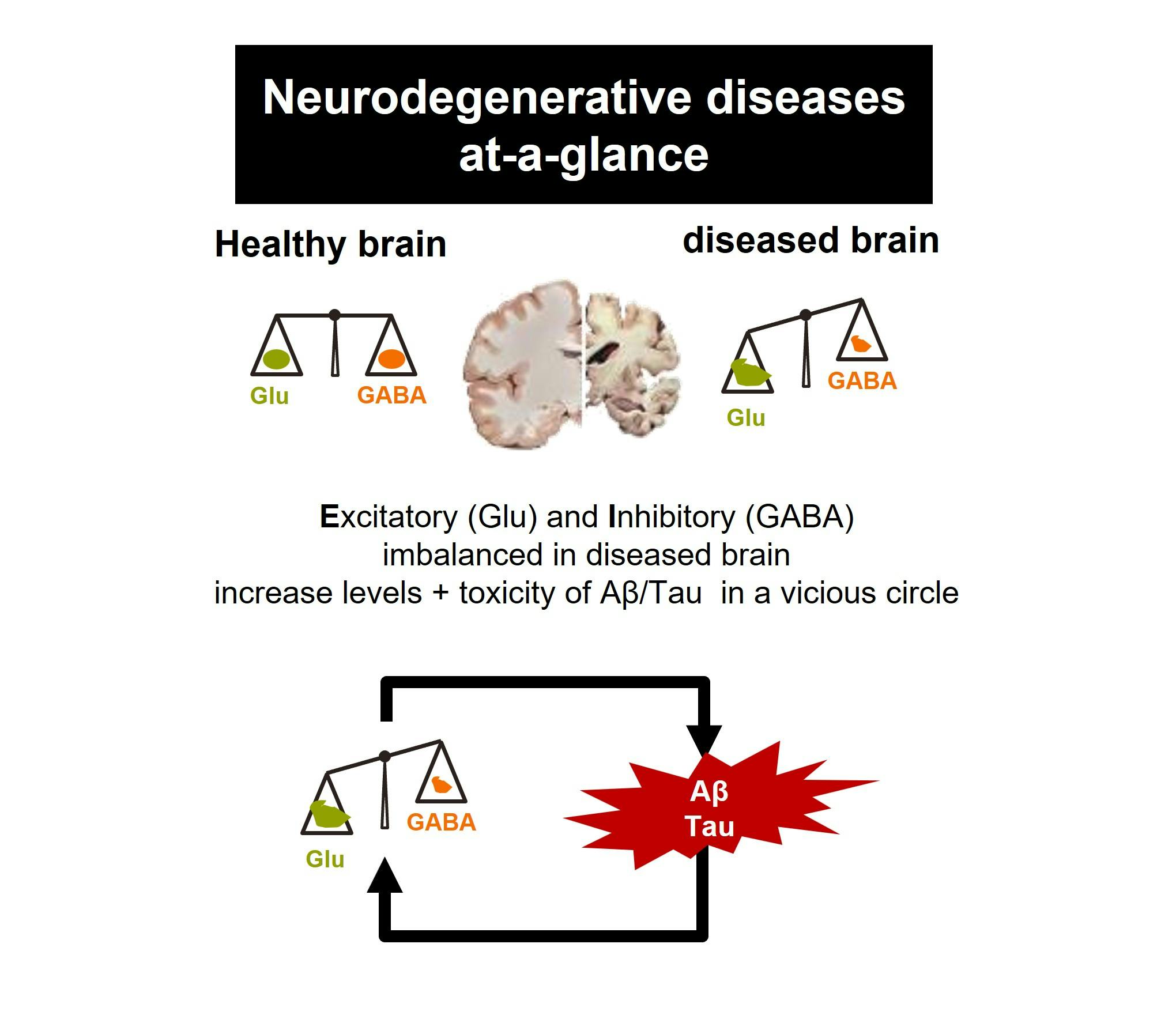
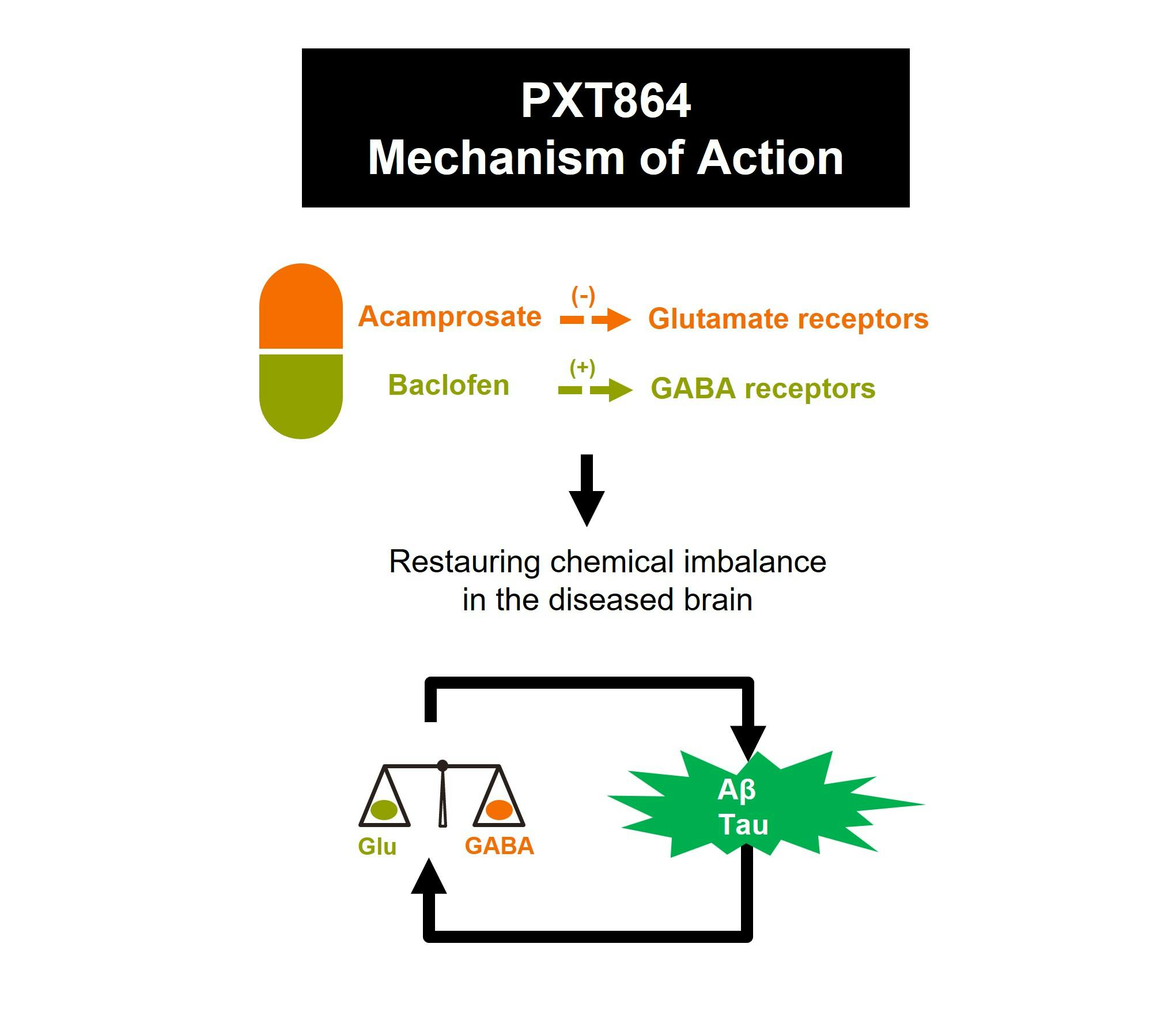
PXT864 is a novel fixed-dose synergistic combination of baclofen and acamprosate formulated as a pill given twice a day. PXT864’s assumed main mechanism of action in neurodegenerative diseases is the restoration of balance between excitatory (glutamate activity) and inhibitory (GABA activity) pathways, disrupted by toxic factors such as Aβ oligomeric peptides in Alzheimer’s disease for instance.
PXT864 showed positive safety and tolerability results in a Phase 1 clinical trial that enrolled 24 healthy volunteers.
PXT864’s most advanced indication was Alzheimer’s disease. An additional Phase 1 clinical trial, which enrolled 20 healthy volunteers, showed initial efficacy in a scopolamine dementia-induced model. PXT864 also generated encouraging results in a Phase 2a clinical trial completed in 2015 in 45 patients with mild Alzheimer’s disease.
In 2023, Pharnext anyway decided to stop the development of PXT864 in Alzheimer's disease despite these encouraging preclinical and clinical results, because remaining insufficient for commercial exploitation.
The overlapping genetic, molecular, and cellular characteristics of Alzheimer’s disease and Amyotrophic Lateral Sclerosis (ALS) supported investigating the potential effectiveness of PXT864 in this latter indication.
PXT864 has generated encouraging results in several preclinical ALS models (Boussicault et al. J Neurosci Res. 2020) :
• significant inhibition of synergistic loss of neuromuscular junctions between neuronal and muscle cells;
• protection of the neural network;
• synergy with riluzole and enhancement of its protective effect.
Based on previous Phase 1 safety and tolerability results, PXT864 alone or associated with already approved drugs in ALS is ready to start its clinical development in Phase 2a clinical trial.
Amyotrophic Lateral Sclerosis (ALS) is an orphan, chronic neurodegenerative disease characterized by progressive loss of central and peripheral motor neurons, rapidly leading to major impairment/disability and ultimately to death. ALS affects around 400,000 people worldwide.
There is currently no therapy impacting the course of ALS. The only approved treatment are riluzole and eadaravone, but their efficacy is modest and short-lasting. They have very limited impact on life expectancy and don’t improve quality of life.